Biocontrol is an area of increasing focus for termite control. Below is a summary of the latest research in the subject of biocontrol and general termiticide products.
Latest termiticide and biocontrol research
New potential insecticide – isocycloseram
Syngenta has announced a new insecticide in registration: isocycloseram. Although its performance on German cockroaches was highlighted in the announcement, it is a broad-spectrum insecticide. In laboratory studies on both Reticulitermes flavipes and Coptotermes formosanus, worker termites tunnelled into treated soil, indicating the insecticide was nonrepellent, and 100% mortality was achieved between 3-5 days, even in the lowest concentration.1 The paper also reported that the insecticide had passed at least six years in the United States Forest Service field trials.
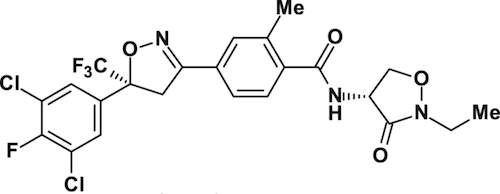
Further evaluation against termites can be expected, with the researchers describing isocycloseram as a non-repellent with delayed effects and activity at low rates, with the potential to be a very effective compound against social insects.
Nanoparticles for Termite Control
Research involving nanoparticles for the control of pests and pathogens in the agriculture sector has been underway for a number of years, but there are few studies on termites. Researchers evaluated nanoparticle solutions of zinc oxide, titanium dioxide and chitosan on the termite Odontotermes formosanus.2 All nanoparticles delivered significant mortality within 48 hours, with effects first being noticed at 24 hours. Titanium dioxide appears to be more toxic to termites than the other nanoparticles, delivering 100% mortality at the two highest levels tested. Interestingly, the nanoparticles themselves were created by mixing the zinc oxide, titanium dioxide or chitosan with a fungus, Scedosporium apiospermum. The researchers believe this research has enormous potential leading to biosynthesised insecticides for termite control with favourable toxicological and environmental safety profiles.
Nanotechnology can also be used to modify the performance of traditional insecticides. Fipronil is a well-known termiticide, but although classed as ‘slow acting’, it still could still benefit from further delayed action. Like many insecticides it also has high toxicity to aquatic life. Researchers have developed a fipronil silica nanoparticle coated with hydroxypropyl cellulose.3 The idea is that the cellulose coating means the fipronil is only activated once inside the termite gut, and the cellulase enzymes break down the cellulose (main picture, above). Trials on Reticulitermes flaviceps demonstrated the mortality of termites exposed to fipronil-loaded nanoparticles increased more slowly than ‘naked’ fipronil, which was expected due to the delayed release, and could possibly improve the transfer effect. Importantly, the cellulose coating also reduced its aquatic toxicity, reducing mortality and improving hatching rates in zebra fish by >6.5 times and 8 times respectively.
The same researchers have also developed metalorganic framework nanoparticles loaded with imidacloprid and capped with sodium lignosulfonate.4 This sodium lignosulfonate layer is degraded in the acidic and laccase-rich environment of the termite gut, releasing the imidacloprid. Compared to imidacloprid in a standard formulation, the nanoparticle exhibits a delayed onset of mortality and activity over a longer period, as the imidacloprid is gradually released.
RNAi Insecticides for Termite Control
A number of physiological processes are triggered by gene expression within termites that help detoxify insecticides. Similarly, genes also control termite behaviours that help with colony immunity, such as allogrooming. If the expression of these genes can be restricted or eliminated, it may provide new routes to control termites. This is called RNAi or RNA interference technology.
The first part of such studies is to identify the genes involved in the process, before utilising RNAi to prevent gene expression. Researchers evaluating the effect of RH-5849 (an IGR), found two genes in Odontotermes formosanus that were involved in the detoxification of RH-5849. Using RNAi, which delivered interference efficiency at 85.3% and 59.59% for the two genes, the lethality of RH-5849 was significantly increased.5
Allogrooming is a key element in helping maintain colony health by removing potential pathogens. Understanding the behavioural and physiological processes around allogrooming could therefore identify opportunities to influence this behaviour and open up new options for termite control, especially with reference to biopesticides.
One group working on Coptotermes formosanus has identified 60 micro RNAs (genes) that are expressed when the termites are exposed to the pathogenic fungus Metarhizium anisopliae.6 Some of the changes in gene expression can have positive effects (helping the termite fight infection) whereas some can have negative effects. It appears that the fungus is manipulating gene expression in the termite to help it infect the termite. If the expression of these genes can be manipulated to make the termites more susceptible to fungal pathogens, it could potentially be used as a control method.

miR-571 is significantly upregulated in termites infected with M. anisopliae. This upregulation appears to reduce the normal termite pathogen avoidance and grooming behaviours – an example of a pathogen influencing the gene expression in the termite to its advantage. The researchers demonstrated that by exposing termites to a miR-572 agomir (either through injection or ingestion), miR-571 upregulation was further increased, resulting in significantly increased mortality in M. anisopliae-infested termites.
Working with Odontotermes formosanus, researchers have identified an odorant receptor co-receptor (Orco) that was expressed when the termites were exposed to SM1 (Serratia marcescens), a gram-negative bacteria with insecticidal properties. This gene expression resulted in increased allogrooming.7 Subsequent interference in the expression of the Orco resulted in reduced allogrooming and increased mortality in termites exposed to SM1. The researchers believe this finding could lead to the integration of RNA interference (RNAi) with biopesticides to advance research on termite RNA biopesticides.
References
1 Saran, R.K. et al. (2025) ‘Efficacy and utility of isocycloseram a novel isoxazoline insecticide against urban pests and public health disease vectors’, Pest Management Science, 81(2), pp. 978–989. https://doi.org/10.1002/ps.8497
2 Nasser, R., Ibrahim, E., Fouad, H., Ahmad, F., et al. (2024) ‘Termiticidal Effects and Morpho-Histological Alterations in the Subterranean Termite (Odontotermes formosanus) Induced by Biosynthesized Zinc Oxide, Titanium Dioxide, and Chitosan Nanoparticles’, Nanomaterials, 14(11). https://doi.org/10.3390/nano14110927
3 Wu, Z. et al. (2024) ‘Cellulase-responsive hydroxypropyl celluloseanchored hollow mesoporous silica carriers for pesticide delivery’, International Journal of Biological Macromolecules, 277(4) https://doi.org/10.1016/j.ijbiomac.2024.134612
4 Wu, Z. et al. (2025) ‘Sodium-lignosulfonate-conjugated metal-organic frameworks as dual-stimulus-responsive carriers for improved pesticide targeting’, International Journal of Biological Macromolecules, 305(2). https://doi.org/10.1016/j.ijbiomac.2025.141275
5 Xiong, Y., Xie, L. and Tang, F. (2025) ‘Silencing OdfoP-gp1 and OdfoP-gp3 with dsRNA increased the lethality of the non-steroidal ecdysone agonist RH-5849 against Odontotermes formosanus (Shiraki)’, Pesticide Biochemistry and Physiology, 208 https://doi.org/10.1016/j.pestbp.2024.106226
6 Chen, W. and Li, Z. (2025) ‘miR-571 manipulating termite immune response to fungus and showing potential for green management of Coptotermes formosanus (Blattodea: Isoptera)’, Pesticide Biochemistry and Physiology, 208. https://doi.org/10.1016/j.pestbp.2024.106274
7 Lu, X. et al. (2024) ‘The function of OforOrco in the allogrooming behavior of Odontotermes formosanus (Shiraki) induced by Serratia marcescens Bizio (SM1)’, Pesticide Biochemistry and Physiology, 204. https://doi.org/10.1016/j.pestbp.2024.106004


